We have an on-going programme to explore applications of optical coherence tomography (OCT) in embryology, beginning with a BBSRC grant which ran from 2006 to 2009, continuing in 2017 with the use of speckle variance OCT to monitor embryo viability, and then receiving further BBSRC funding for 5DHiResE project beginning in 2019.
Developmental biology is moving away from standard antibody staining of fixed embryos toward live imaging, which can provide novel insights into various known developmental events. However, live imaging is restricted to the embryo surface. In transparent and semi-transparent organisms amenable to transgenic manipulation the expression of some fluorescent proteins can be detected by laser scanning confocal microscopy. Although by expressing fluorescent proteins one can successfully image transparent areas, effective imaging at depth in embryos and tissue is still restricted.
From 2006 to 2009, Prof Adrian Podoleanu held a BBSRC grant with Dr Jim Bloor from the School of Biosciences, employing Dr Adrian Bradu and Dr Lisha Ma (Biosciences) to investigate the potential of OCT-based measurement on well-characterised and novel animal models for embryology and cell imaging. The aims of the joint research activity were:
- to develop an OCT platform for high resolution imaging of cells and embryos, in particular a dual en-face/Doppler OCT instrument capable of recording multiple aspects of heart activity, including heart contraction cycle dynamics, ostia dynamics, heartbeat rate and rhythm, speed of heart wall movement and light reflectivity of cardiomyocytes in situ. We successfully demonstrated the capability of such an instrument by characterizing in detail cardiac activity in genetic models for heart disease in Drosophila melanogaster.
- to combine the imaging depth capability of OCT with the high lateral resolution of fluorescent imaging by developing a dual OCT-confocal microscope. We demonstrated a combined instrument of the two imaging modalities (Spectral Domain OCT and laser scanning fluorescence microscopy (LSFM)) to acquire information on cardiac function in larval Drosophilamelanogaster. Our dedicated imaging instrument is able to sequentially provide cross-sectional OCT and C-scan LSFMimages. With this dual-imaging system, the heart can be easily located and visualized within the specimen and the change of the heart shape in a cardiac cycle can be monitored.
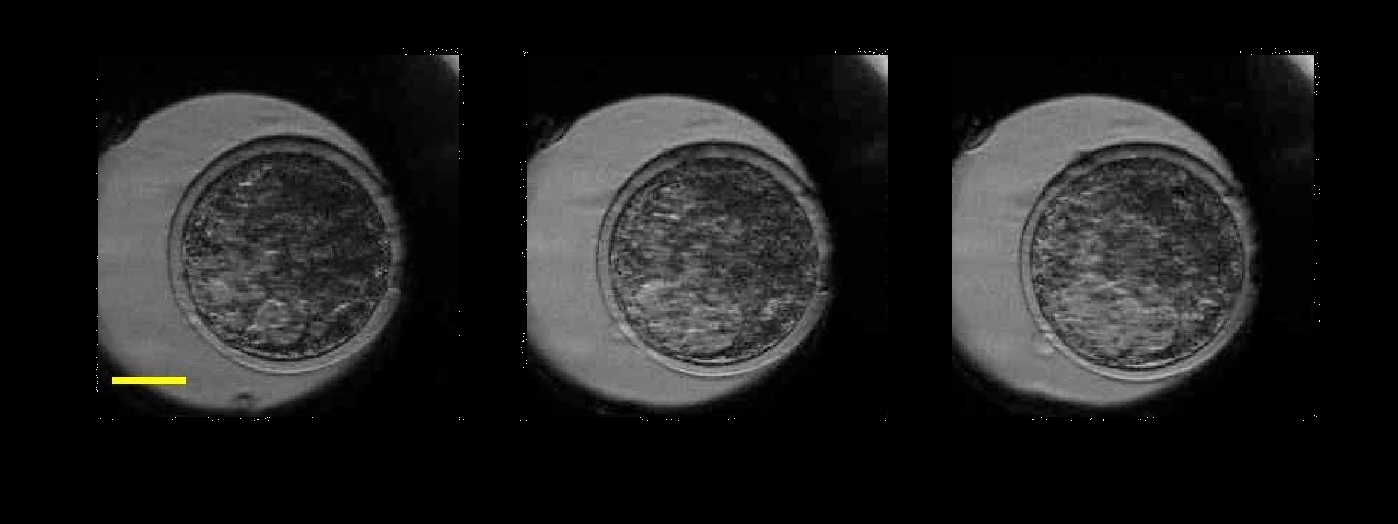
In 2017, an on-going collaboration with the School of Biosciences led to the demonstration of the use of speckle variance OCT for assessment of the viability of bovine embryos. Following IVF, the embryos were monitored for up to 18 hours by OCT and speckle variance OCT to monitor the micron-scale movements which are potentially associated with viability. The results, obtained as part of the PhD of Sophie Caujolle and published in Biomedical Optics Express, show that speckle variance OCT could provide a a fast, non-invasive way to measure the levels of damage sustained by an embryo, for example after cryopreservation.
In 2019 we began the 5DHiResE project, researching the use ofnon-invasive, non-destructive imaging techniques, such as Optical Coherence Tomography (OCT) to establish the vitality of in-vitro produced (IVP) embryos (both bovine and porcine). This project is the result of across-disciplinary collaboration between the AOG and the Prof Griffin’s lab in the School of Biosciences. The project is supported by external partners, ActIVF-ET, Care Fertility Group, and Topigs Norsvin.
Publications
- Caujolle, S., Ramona Cernat, G. Silvestri, M. J. Marques, Adrian Bradu, T. Feuchter, G. Robinson, Darren K. Griffin, and A. Podoleanu. “Speckle variance OCT for depth resolved assessment of the viability of bovine embryos.” Biomedical optics express 8, no. 11 (2017): 5139-5150.
- L. Ma, A. Bradu, A. Gh. Podoleanu, J. Bloor, Arrhythmia Caused by a Drosophila Tropomyosin Mutation Is Revealed Using a Novel Optical Coherence Tomography Instrument, PLoS ONE 5(12): e14348. doi:10.1371/journal.pone.0014348 (2010).
- L. Neagu, A. Bradu, L. Ma, J. Bloor, and Adrian Gh. Podoleanu, “Multiple-depth en face optical coherence tomography using active recirculation loops,” Opt. Lett. 35, 2296-2298 (2010).
- A. Bradu, Lisha Ma, J. Bloor and A. Podoleanu, Dual optical coherence tomography/fluorescence microscopy for monitoring of Drosophila melanogaster larval heart, J. Biophotonics, Opt., Vol. 2(6-7), p.380-388, 2009.
- A. Bradu, L. Ma, J. Bloor, A. Gh.Podoleanu, Sequential en-face optical coherence tomography imaging and monitoring of Drosophila Melanogaster larval heart, in Optical Coherence Tomography and Coherence Domain Optical Methods in Biomedicine XIII, edited bys G. Fujimoto, Joseph A. Izatt, Valery V. Tuchin, Proceedings of SPIE Vol. 7168 (SPIE, Bellingham, WA 2009).
- A. Bradu, L. Ma, J. Bloor, A. Gh. Podoleanu, Using en-face optical coherence tomography to analyse gene function in Drosophila Melanogaster larval heart in 1st Canterbury Workshop on Optical Coherence Tomography and Adaptive Optics, edited by Adrian Podoleanu, Proceedings of SPIE Vol. 7139 (SPIE, Bellingham, WA 2008).
- A. Bradu, L. Ma, J. Bloor, A. Gh. Podoleanu, Combining confocal microscopy and optical coherence tomography for imaging in developmental biology, in Biophotonics: Photonic Solutions for Better Health Care, edited by Jurgen Popp, Wolfgan” Drexler, Valery V. Tuchin, Dennis L. Matthews, Proceedings of SPIE Vol. 6991 (SPIE, Bellingham, WA 2008).
- A. Bradu, L. Ma, J. Bloor, A. Gh. Podoleanu, Versatile confocal/optical coherence tomography system for embryonic developmental imaging, in Coherence Domain Optical Methods and Optical Coherence Tomography in Biomedicine XII, edited by Joseph A. Izatt, James G. Fujimoto, Valery V. Tuchin, Proceedings of SPIE Vol. 6847 (SPIE, Bellingham, WA 2008).