Lead Investigators: Prof. Dan Bracewell and Dr Lalintip Hocharoen
Domestic manufacture of biopharmaceuticals in Thailand would reduce drug costs, improving accessibility to patients. However, the cost of goods for biopharmaceuticals remains high because of the expensive downstream operations currently employed in their production. The challenge of affordability for a broad population in Asia is to reduce these costs by design of processes with improved economics while still being able to make the case for “biosimilarlity”.
This part of the project concerns the development of strategies for cost-effective downstream processing for biosimilar products. It will involve building process development expertise in Thailand and focusing on methods to scale-down these activities to rapidly explore new process options. This will benefit from Prof Bracewell’s experience in scale-down mimics of unit operations with appropriate analytical methodology to achieve new levels of process understanding.
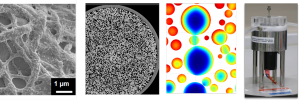
Images (L to R): i) Nanofibre based adsorbent binding viral vector products (adenovirus) ii) X-ray computed tomography image of the packing of chromatography column iii) visualisation of a chromatography modelling describing protein adsorption at the bead level iv) solid-liquid shear device to investigate therapeutic protein stability during manufacturing.
